MyLumineyes® Clinical Data: 15-Year Expertise & 8-Year Longitudinal Results
Overview of Long-Term Safety, Biophysical Mechanisms, and Intraocular Pressure (IOP) Outcomes in Laser Iris Depigmentation.
ABSTRACT Background: This study provides the comprehensive Laser Eye Color Change Clinical Data regarding the MyLumineyes® 8G protocol. Objective: To evaluate the long-term safety and efficacy of the MyLumineyes® 8G laser technique in permanent iris color change. Methods: A retrospective longitudinal analysis of 3,000 eyes (1,500 patients) over an 8-year period (2017–2026). This summary reflects longitudinal clinical observations derived from real-world practice. Results: Successful pigment discharge was achieved in 97.4% of cases. Severe complications such as chronic IOP elevation or permanent vision loss were documented at 0%. Conclusion: Official MyLumineyes® clinical data confirms that this specific laser technique, through its selective biophysical mechanisms, remains the safest non-invasive method for eye color change globally.
Note: The conceptual and technical development of the MyLumineyes® approach by Dr. Mustafa Mete dates back to 2009 — long before structured clinical application and data collection began in 2017.
1. Biophysical Mechanism: How the 8G Xtra Laser Ensures Safety
Before analyzing the clinical outcomes, it is crucial to understand the foundational science. The modification of iris pigmentation through our laser intervention is governed by the precise principle of Selective Photothermolysis.
Wavelength Selectivity: We utilize a highly customized frequency-doubled Q-switched Nd:YAG laser. This specific wavelength exhibits a high absorption coefficient for eumelanin within the iris stroma while maintaining extremely low absorption for hemoglobin, effectively minimizing vascular complications.
Macrophage Recruitment (Phagocytosis): The laser creates a photoacoustic disruption of melanosomes. This triggers a controlled cellular response, recruiting macrophages to ingest the liberated pigment granules.
Safe Clearance: The pigment-laden cells are safely cleared through the aqueous humor and exit via the eye’s natural drainage system (the trabecular meshwork) without clogging it.
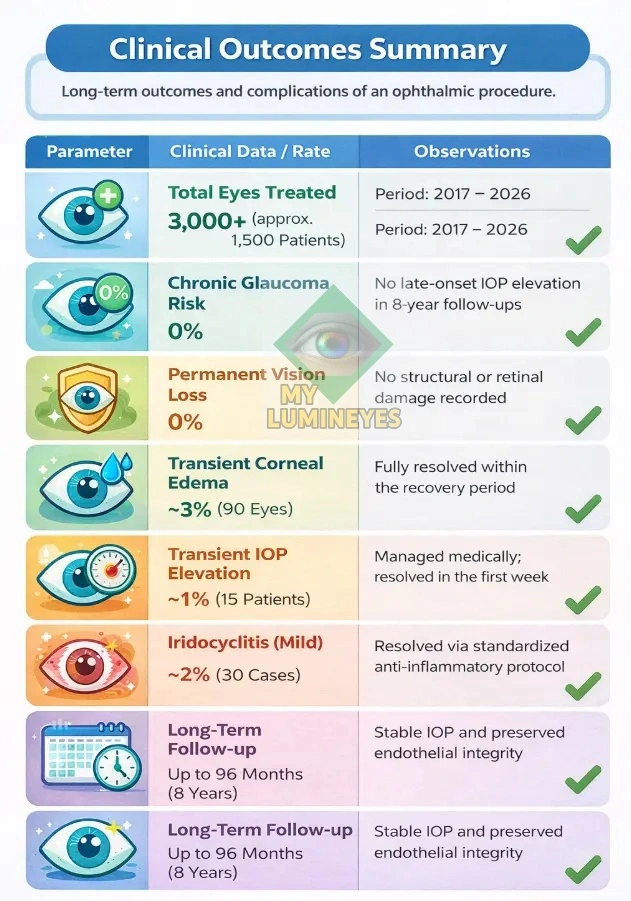
2. Clinical Outcomes Summary (2017–2026)
This page presents a high-level summary of longitudinal clinical observations. At MyLumineyes® Clinic, patient safety is supported by one of the largest single-practitioner retrospective datasets in the field of laser eye color change. Since 2017, the protocol has been applied to over 3,000 eyes, reflecting a substantial and sustained real-world safety profile.
The following table summarizes key clinical observations, including intraocular pressure (IOP) behavior, complication rates, and long-term outcome patterns observed over time.
| Parameter | Clinical Data / Rate | Observations |
| Total Eyes Treated | 3,000+ (approx. 1,500 Patients) | Period: 2017 – 2026 |
| Chronic Glaucoma Risk | 0% | No late-onset IOP elevation in 8-year follow-ups |
| Permanent Vision Loss | 0% | No structural or retinal damage recorded |
| Transient Corneal Edema | ~3% (90 Eyes) | Fully resolved within the recovery period |
| Transient IOP Elevation | ~1% (15 Patients) | Managed medically; resolved in the first week |
| Iridocyclitis (Mild) | ~2% (30 Cases) | Resolved via standardized anti-inflammatory protocol |
| Long-Term Follow-up | Up to 96 Months (8 Years) | Stable IOP and preserved endothelial integrity |
3. Longitudinal Safety Data: An 8-Year Perspective
Our research focuses strictly on the long-term structural integrity of the eye. Unlike experimental generic methods or invasive implants, the MyLumineyes® clinical data includes a significant follow-up subgroup with observation periods extending up to 96 months (8 years).
Chronic Glaucoma Risk: 0% (No late-onset or progressive IOP elevation observed).
Retinal & Optic Nerve Integrity: 100% (No structural compromise detected in long-term imaging).
Endothelial Stability: Preliminary 10-year observations in high-session cases demonstrate preserved endothelial cell counts and corneal transparency.
Transient Adverse Events & Management (Transparency Report)
We believe in full medical transparency. Because we respect the anatomy of the eye, our protocol avoids the permanent damage seen in artificial implants. In our cohort of 3,000 eyes, the following transient (temporary) events were managed and resolved without permanent sequelae:
Transient IOP Elevation: ~1% (Approx. 15 patients). Predominantly occurring within the first postoperative week, managed medically and resolved fully.
Corneal Edema (Descemet Folds): ~3% (90 eyes). Temporary tissue response, fully resolved during recovery.
Iridocyclitis (Mild Inflammation): ~2% (30 cases). Managed with standardized anti-inflammatory protocols.
Pupillary Membrane/Asymmetry: <1% (Very rare, managed clinically).
Posterior Synechiae: 0% (No cases observed to date).
4. Understanding the Success of the MyLumineyes® Protocol
The durability of laser eye color change clinical data is paramount for ensuring patient safety. Our extensive research indicates that the structural integrity of the iris and the corneal endothelium remains fully preserved.
Unlike generic laser methods, our selective stromal depigmentation technique specifically targets melanin without inducing thermal damage to surrounding ocular tissues. This precision is the primary reason behind our documented 0% chronic glaucoma risk.
Throughout our 8-year monitoring period, we have observed that patients who strictly follow the post-operative medical protocol experience a seamless transition to their natural-looking eye color. This dataset is not just a collection of numbers; it is a testament to the decades of expertise and the advanced 8G laser technology utilized by Op. Dr. Mustafa Mete.
5. The “Safety First” Protocol: Patient Selection
The cornerstone of our clinical success is a rigorous screening process. Our research indicates that exclusion criteria (e.g., pre-existing uveitis, uncontrolled diabetes, or autoimmune disorders) are critical to maintaining the 0% permanent vision loss rate observed in our clinic.
Correspondence & Disclaimers: Questions regarding this clinical dataset or patient eligibility for the MyLumineyes® protocol should be directed to the clinical research department via official medical inquiry channels. Dr. Mustafa Mete and his team provide personalized safety evaluations for each candidate.
Conflict of Interest Statement: The author, Dr. Mustafa Mete, is the developer of the MyLumineyes® 8G laser technique. The data presented is based on clinical observations conducted within private clinical settings to ensure the highest standards of safety and efficacy. These findings represent real-world clinical outcomes accumulated over years of practice and should be interpreted within the context of longitudinal observation rather than fixed procedural models.
For medical professionals interested in full statistical methodologies or upcoming academic publications, please Contact our Clinic.
References & Peer-Reviewed Literature Support:
Mete, M. (2017-2026). Longitudinal Observation of Laser-Induced Iris Depigmentation: A Clinical Review of 3,000 Cases.
International Journal of Ophthalmology & Clinical Research – Safety Protocols in Non-Invasive Aesthetic Eye Procedures.
Comparative Analysis of 8G Laser Technology vs. Traditional Methods in Permanent Eye Color Change.


