Off-Label Use in Laser Eye Color Change: Clinical Context and Safety Boundaries
Laser eye color change represents a sophisticated application within modern ophthalmology, where established laser systems are utilized beyond their initial regulatory indications. This approach is defined as off-label use—a recognized and integral part of medical practice when guided by expert clinical judgment and evidence-based protocols.
Understanding Off-Label Application in Ophthalmic Lasers
In this context, off-label use does not imply experimental uncertainty. Instead, it reflects the advanced adaptation of Selective Photothermolysis technology to a specific biological target: the iris stroma. While these laser systems are designed for precise ocular interaction, their application in iris depigmentation requires a specialized modification of energy delivery, targeting depth, and pulse sequencing.
Key Insight: The distinction lies not in the device itself, but in the proprietary methodology governing its clinical application.
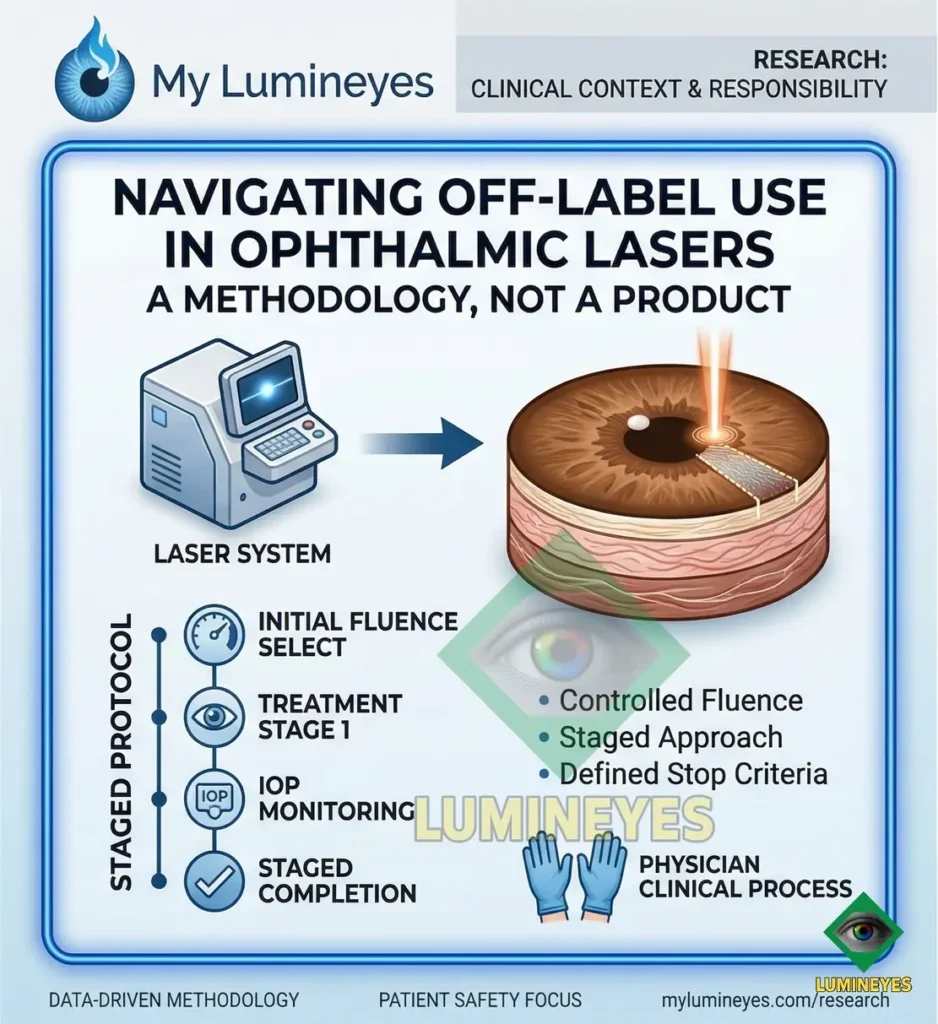
Protocol-Dependent Safety
Unlike standardized surgeries, the safety profile of laser iris depigmentation is not an intrinsic property of the laser hardware, but of the clinical protocol. Key safety determinants include:
- Fluence & Spot Size: Precise selection to prevent deep stromal penetration.
- Staged Planning: Preventing acute pigment overload in the Trabecular Meshwork.
- IOP Monitoring: Defined intervals to track intraocular pressure dynamics.
- Biological Stop Criteria: Ceasing treatment based on individual tissue response.
The Necessity of Clinical Judgment
This procedure transforms from a device-based intervention into a physician-dependent clinical process. Decision-making extends to patient selection and the interpretation of subtle biological responses, ensuring that Mylumineyes safety standards are maintained throughout the transition.